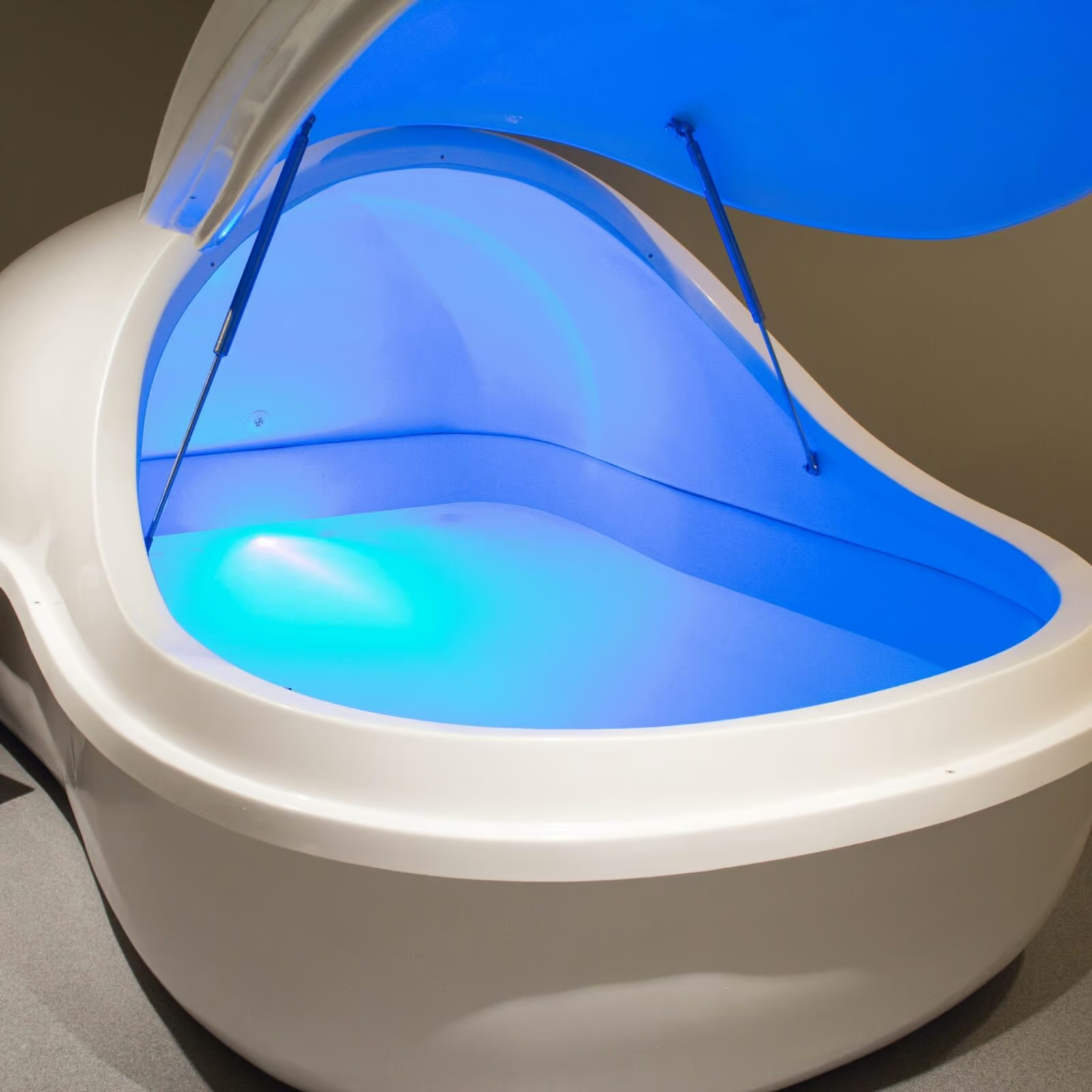
Royal Spa has been engineering water therapy equipment since 1981. Epsom Salt Capable since 1995. Float Tanks since 2012.
We know what holds up — and what doesn’t.
We build the equipment and sell it direct. No distributor markup, no middleman, and a 5-year structural warranty behind every float tank we make. Four configurations from commercial float pods and pools to a personal tank you can put in a spare bedroom. Tell us what you’re building and we’ll tell you which one fits.
Float Therapy as a Revenue Stream
Float therapy commands $75–$89 per 60-minute session with near-zero variable cost once the equipment is paid for. Water, salt, electricity — a few dollars per float. Most established float centers reach equipment payback within 3–5 years.
The question business owners ask us isn’t “does floating work.” It’s “which tank fits my space, my volume, and my clients?” That’s exactly the conversation our specialists have every week — with spa owners, chiropractors, PT clinics, gyms, and dedicated float centers.
With benefits that last
Royal Spa®
Float Tank Warranty
Frequently asked questions
How many float sessions can one tank handle per day?
Most commercial operators run 3–5 sessions per tank per day at standard 60-minute sessions with 30-minute turnovers for filtration cycling and room prep. Four sessions per tank per day is a reliable planning number for most operations.
What’s the difference between a float pod and a float room?
A float pod is an enclosed tank — the traditional sensory deprivation form factor. A float room is an open-top design where clients walk in through a door rather than lifting a lid. Open rooms eliminate claustrophobia concerns entirely, which can meaningfully widen your client base. Many multi-tank operators offer both.
How long does a float tank last?
With proper maintenance, a well-built float tank should last 15–20+ years. Mechanical components — pumps, heaters, filtration — will need periodic replacement. The shell itself, properly constructed and maintained, is essentially permanent. Our 5-year structural warranty reflects that confidence.
What does it cost to run a float tank per month?
Variable operating costs per tank typically run $200–$500 per month depending on local energy rates, water costs, and salt top-off frequency. This makes float therapy one of the lowest variable-cost wellness services available — which is why the margins are attractive for operators.
What’s the difference between a residential and commercial float tank?
Commercial tanks are built for higher daily throughput, faster filtration turnover between sessions, and more robust component specs. Royal Spa’s Float Pod and Float Room XL are designed for commercial use. The Personal Float Tank is purpose-built for home use — more economical, simpler installation, designed for 1–2 sessions per day.
Do you sell direct, or through dealers?
Direct from the manufacturer. No distributor markup, factory-level support, and our 10% price-beat guarantee. You also get ongoing support from the same team that built your tank — not a third-party dealer who resells whatever brand gives them the best margin.